Biosafety Protocol News - Issue 14, 2020
 |
|
Inspiring Quotes:
20th anniversary of the adoption of the Cartagena Protocol on Biosafety and
10th anniversary of the Nagoya Kuala - Lumpur Supplementary Protocol on Liability and Redress |
-Elizabeth Maruma Mrema, Executive Secretary, the Secretariat of the Convention on Biological Diversity, the United Nations Environment Programme (UNEP)
 |
|
The immense advances in biotechnology and the speed of these developments mean the Cartagena Protocol is more relevant than ever. The development of the post-2020 global biodiversity framework and the Implementation Plan for the Cartagena Protocol constitute a crucial opportunity for countries to make commitments in this regard.
The importance of rules on liability and redress for damage to biodiversity resulting from LMOs is reflected in the Supplementary Protocol on Liability and Redress. I urge and invite all Parties to the Biosafety Protocol yet to do so to consider ratifying the Supplementary Protocol. |
Elizabeth Maruma Mrema appointed head of UN Biodiversity Convention, June 2020 International biosafety treaty marks its twentieth anniversary Nagoya-Kuala Lumpur Supplementary Protocol on Liability and Redress celebrates tenth anniversary
-Galina Mozgova, Biosafety Clearing-House Focal Point, Belarus
 |
|
Belarus recognizes the importance of intersectoral collaboration among all Parties in the field of biosafety to implement measures aimed to contribute to ensuring an adequate level of protection in the field of the safe transfer, handling and use of living modified organisms (LMOs). In 2019, a new version of the law was adopted: “On the Safety of Genetic Engineering Activities”. Projects were also carried out with the support of the Global Environment Facility and Central European Initiative, aimed at educating and training key stakeholders and consolidating cooperation for the implementation of the Protocol at the national, regional and global levels.
В Республике Беларусь признается важность межсекторального взаимодействия между всеми сторонами в области биобезопасности для выполнения мер, направленных на предотвращение или снижение до безопасного уровня возможных вредных воздействий ЖИО. В 2019 г. была принята новая редакция закона «О безопасности генно-инженерной деятельности». В 2018-2019 гг. осуществлялось выполнение проектов при поддержке Глобального Экологического Фонда и Центральной Европейской Инициативы, направленных на просвещение, обучение всех ключевых сторон и консолидацию сотрудничества для реализации Протокола на национальном, региональном и глобальных уровнях. |
-National Focal Point for the Cartagena Protocol on Biosafety, China
 |
|
China has issued more than 10 biosafety-related laws and regulations to address the potential risks of modern biotechnology and its products to human health, the environment and biodiversity. In 2019, the 4th national report of the Cartagena Protocol on Biosafety was completed by the Ministry of Ecology and Environment in collaboration with nine departments. The Biosecurity Law of People’s Republic of China was adopted in October 2020 and will come into effect on April 15, 2021. The Biosecurity Law is formulated in a bid to provide the legal basis and guarantee for maintaining national security, preventing and responding to biosecurity risks, safeguarding people's life and health, protecting biological resources and the ecological environment, promoting the sound development of biotechnology and the building of a community with a shared future for mankind, and achieving the harmonious coexistence of mankind and nature.
为了防范现代生物技术及其产品对人类健康、环境及生物多样性构成的潜在风险,中国发布实施了10余项生物安全相关的法律法规。2019年,生态环境部会同9个部门共同编制了《卡塔赫纳生物安全议定书》第四次国家报告。2020年10月,《生物安全法》获得通过,并将于2021年4月15日起施行。该法为维护国家安全,防范和应对生物安全风险,保障人民生命健康,保护生物资源和生态环境,促进生物技术健康发展,推动构建人类命运共同体,实现人与自然和谐共生,提供了法律依据和保障。 |
-Mohammed Mehdi, National Focal Point for the Cartagena Protocol on Biosafety, Morocco
 |
|
Morocco notes that biosafety issues are of high importance. The national focal point of the Cartagena Protocol on Biosafety, the focal point for the Biosafety Clearing-House and the national competent authority have been appointed since 2011. Recently, the Department of the Environment has finalized an action plan for the implementation of the Protocol that provides a road map for all stakeholders.
Le Maroc note que les questions de biosécurité sont d'une grande importance. Le point focal du Protocole, le point focal du Centre d’échange pour la prévention des risques biotechnologiques ainsi que l'Autorité Nationale Compétente ont été désignés depuis 2011. Récemment, le Département de l'Environnement a finalisé un plan d'action de mise en œuvre du Protocole qui constitue une feuille de route pour l'ensemble des parties prenantes.
يلاحظ المغرب أن قضايا السلامة الأحيائية لها أهمية عالية. تم تعيين نقاط الاتصال الوطنية للبروتوكول وغرفة تبادل معلومات السلامة الأحيائية والسلطة الوطنية المختصة منذ سنة 2011. وضع قطاع البيئة اللمسات الأخيرة على خطة عمل لتنفيذ البروتوكول والتي تشكل خارطة طريق لجميع أصحاب المصلحة. |
-Casper Linnestad, National Focal Point for the Cartagena Protocol on Biosafety, Norway
 |
|
The Norwegian Gene Technology Act from 1993, emphasizes that the deliberate release of LMO’s should have no detrimental effect on health nor the environment. Norway welcomes the sustainable use of biotechnology in different areas. |
-Jimena Nieto Carrasco, Former Co-Chair Open-Ended Working Group of Legal and Technical Experts on Liability and Redress
 |
|
I would like us to celebrate the Nagoya-Kuala Lumpur Supplementary Protocol, an instrument that originates from the Cartagena Protocol on Biosafety and that establishes international rules and procedures in the field of liability and redress for the potential adverse impact that certain living modified organisms (LMOs) may have on the conservation and sustainable use of biodiversity.
Igualmente quisiera celebrar el Protocolo Suplementario de Nagoya Kuala Lumpur, un instrumento que se origina en el Protocolo de Cartagena y que establece reglas y procedimientos internacionales en el campo de la responsabilidad y la compensación por el eventual impacto adverso que puedan llegar a tener ciertos OVM´s en la conservación y el uso sostenible de la biodiversidad. |
Useful Information
 New Head of Biosafety Unit, Ms. Wadzanayi Mandivenyi New Head of Biosafety Unit, Ms. Wadzanayi Mandivenyi
 SCBD's Action Agenda for commitments from non-state actors. Make a commitment! SCBD's Action Agenda for commitments from non-state actors. Make a commitment!
 Biosafety commitment: Every Women Home Center, Nigeria - Public awareness Biosafety commitment: Every Women Home Center, Nigeria - Public awareness
 Biosafety commitment: The Institute of Genetics and Cytology, Belarus - Detection and Identification Biosafety commitment: The Institute of Genetics and Cytology, Belarus - Detection and Identification
 Capacity-building video on biosafety education Capacity-building video on biosafety education
 The COP-MOP 9 Decisions booklet in all UN official languages
Key notification: UN Biodiversity Convention adopts 2021 interim budget The COP-MOP 9 Decisions booklet in all UN official languages
Key notification: UN Biodiversity Convention adopts 2021 interim budget
Statistics and Other Information
- Cartagena Protocol on Biosafety: Sierra Leone deposited its instrument of accession to the Cartagena Protocol on 15 June, bringing the total number of Parties to 173.
- Nagoya – Kuala Lumpur Supplementary Protocol on Liability and Redress: one instrument of ratification/accession was deposited on 14 April – by Colombia – bringing the total number of Parties to the Supplementary Protocol to 48.
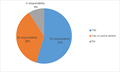 Survey outcome of the Programme of Work on public awareness and participation regarding LMOs Survey outcome of the Programme of Work on public awareness and participation regarding LMOs
Recent and Upcoming Biosafety Events

In response to decision CP-9/3 and CP-9/7, a draft Implementation Plan for the Cartagena Protocol and Capacity-Building Action Plan for the period 2021-2030 was developed to support Parties in their implementation efforts and to facilitate and help develop the strengthening of capacities for implementation.
Following a peer review process of the draft plans (December 2019-January 2020), the Liaison Group on the Cartagena Protocol met online from 20 to 23 April 2020 to review the draft Capacity-Building Action Plan.

A meeting of Ad Hoc Technical Expert Group on Risk Assessment was held online from 30 March to 3 April. The AHTEG considered the needs and priorities for further guidance with regard to specific topics of risk assessment and prepared an analysis and agreed that guidance for risk assessment on LMOs containing engineered gene drives should be developed.

The Informal Advisory Committee (IAC) to the BCH held its eleventh meeting online from 1 to 4 December 2020. It was the IAC’s first meeting in four years, and it agreed on a variety of recommendations regarding improving the functionality of the BCH Central Portal in relation to its migration to the new platform.

The third joint Aarhus Convention/CBD round table on public awareness, access to information and public participation regarding LMOs and GMOs was convened during the reporting period, in Geneva, Switzerland, from 16 to 18 December 2019. A total of 45 participants, including 11 focal points from the Cartagena Protocol and 16 focal points from the Aarhus Convention, and 18 organizations attended the round table. At the meeting, participants shared knowledge, experiences and lessons learned in implementing Article 23 and recommendations for strengthening implementation at the national level. Input was provided to a draft pocket guide on access to information that had been prepared jointly by the Secretariat of the Aarhus Convention and the SCBD. A report of the round table is available at http://www.unece.org/index.php?id=50759.
Upcoming events
Twenty-fourth meeting of the Subsidiary Body on Scientific, Technical and Technological Advice, 2 - 7 January 2021
Third meeting of the Subsidiary Body on Implementation, 9 - 14 January 2021
Tenth meeting of the Conference of the Parties serving as the meeting of the Parties to the Cartagena Protocol on Biosafety, 3 - 15 May 2021, Kunming, China
Editors: Ulrika Nilsson Wadzanayi Mandivenyi
Archive: Previous newsletter issues
Subscription: Newsletter subscription
Disclaimer: The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the Convention on Biological Diversity concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. The views expressed in this publication are those of the authors and do not necessarily reflect those of the two secretariats, nor does citing of trade names or commercial processes constitute endorsement.
|